.
Gaseous Sulfur
Gaseous sulfur refers to sulfur in its gaseous state.
.
Sulfur is a chemical element with the atomic number 16 and is found in many forms, including:
- solid
- liquid
- gas
.
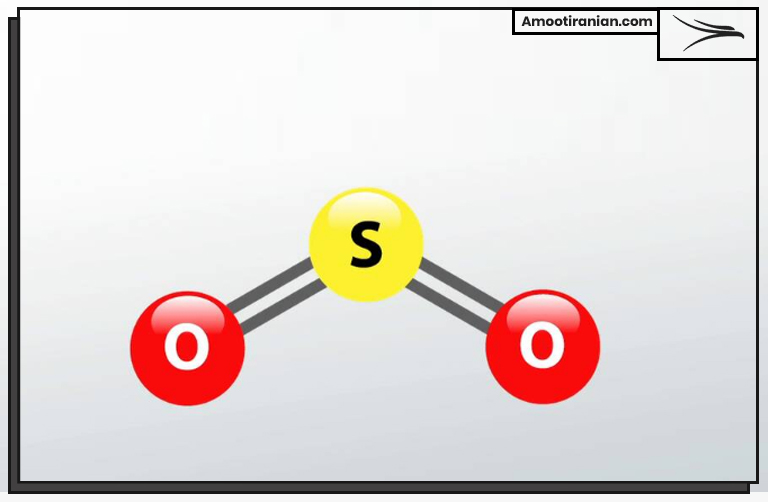
In its gaseous state, sulfur exists as a molecule with two atoms, which is commonly referred to as sulfur gas or sulfur dioxide (SO2).
.

.
Gaseous sulfur is typically produced through the burning of fossil fuels, such as coal and oil, which releases sulfur dioxide into the atmosphere.
It is also produced by volcanic activity and certain industrial processes.
.
Gaseous sulfur has a pungent odor and can be toxic in high concentrations. It is a major contributor to air pollution and can lead to respiratory problems and other health issues.
Sulfur dioxide can also react with other chemicals in the atmosphere to form acid rain, which can have harmful effects on the environment.
.
Gaseous Sulfur Formula
Gaseous sulfur has the same chemical formula as solid or liquid sulfur; S8.
.
.
This formula for gaseous sulfur (S8) means that it is made up of eight sulfur atoms bonded together in a ring.
This form of sulfur is commonly known as elemental sulfur or simply sulfur.
At room temperature and pressure, it is a yellow solid, but it can easily vaporize to form a gas composed of these S8 molecules.
.
Is Gaseous Sulfur Used As A Raw Substance in Medical Applications?
Gaseous sulfur (S8) is not commonly used as a raw substance in medical applications.
.
However, some sulfur compounds are used in medicine, such as sulfonamides, which are antibiotics used to treat bacterial infections.
These sulfur compounds made from gaseous sulfur can be used in medical applications.
.
For example, sulfonamides, which are a class of synthetic antibiotics, are derived from sulfur and have been used to treat bacterial infections since the 1930s.
Sulfur dioxide, which is produced by burning sulfur, is also used in the production of some pharmaceuticals.
.
However, it’s important to note that not all sulfur compounds are suitable for use in medicine.
Some sulfur compounds are toxic or have harmful side effects and are therefore not used in medical applications.
The safety and efficacy of any sulfur compound used in medicine depend on its specific chemical properties and the intended use.
